Ricochet is the best place on the internet to discuss the issues of the day, either through commenting on posts or writing your own for our active and dynamic community in a fully moderated environment. In addition, the Ricochet Audio Network offers over 50 original podcasts with new episodes released every day.
 Learning the Basics and Things that Matter
Learning the Basics and Things that Matter
 To the sadness of all, anonymous has decided to end his amazing Saturday Night Science series (though he’s still writing for Ricochet on similar topics). Out of a sense of both depression and a desire to turn that feeling into something constructive I have decided to write a primer on something which I have found to be important in my life. I want to start out with describing a small toolbox of ideas that is taught to engineers and scientists as a means of looking at the world.
To the sadness of all, anonymous has decided to end his amazing Saturday Night Science series (though he’s still writing for Ricochet on similar topics). Out of a sense of both depression and a desire to turn that feeling into something constructive I have decided to write a primer on something which I have found to be important in my life. I want to start out with describing a small toolbox of ideas that is taught to engineers and scientists as a means of looking at the world.
Of course, there is a lot of confusion about what it is that engineers do. The truth of the matter is that there isn’t just one thing that engineers do; our professional lives are so varied and specialized that the term “Engineer” even with modifiers such as “Chemical,” “Civil,” “Mechanical,” or “Electrical” cannot capture the breadth of depth of what people do in the various disciplines. However, there are typically a few core courses which bind all of the branches of engineering together at the root of their undergraduate training. One of the most important courses which all well-trained engineers take is Thermodynamics.
Thermo gets a bad rap from the outside world. Its very name is scary, foreign, and oftentimes associated with the incomprehensible. So what is thermodynamics if it isn’t a totally baffling subject best left to boffins?
In short, Thermodynamics is the study of how energy and matter interact and the forces they consequently exert in the physical world. It is, of course, impossible to boil-down such a complex and important topic to a Ricochet post, but for those who are interested, the above link does a good job of putting flesh on the bones that I’m seeking to construct here.
There are four laws of Thermodynamics, of which only two are genuinely important for the layman. These laws explain the vast majority of the physical processes and interactions that we see in the world on a day-to-day basis (we will ignore the Zeroth and Third laws here, as they are not at the heart of my intent here).
The First Law of Thermodynamics states simply that Matter and Energy are conserved. This seems fairly straightforward, and when you add in the realization (via Einstein’s famous E=mC2) that matter and energy are simply two sides of the same coin, the straightforwardness becomes even more obvious.
Why is this important? The mathematical implication of this is simple: you can use this principle to draw an imaginary boundary around a system you want assess whether or not claims being made in its name are feasible. For instance, if a traveling salesman wants to sell you a device which can power your home using nothing but the very simple and practical box in his briefcase which produces electricity out of thin air using the Kirlian effect (“Energy companies hate him… See the video before Obama bans it!”) you should not believe this man.
It’s very difficult for something to appear out of nothing, and work (a very specific form of energy) doesn’t poof into existence any more than gold or water do.
In case the First Law is old hat to you, there’s always the Second Law of Thermodynamics. There are several ways to describe it but — in its simplest form — it states that a cold cup of coffee left on a kitchen counter will not spontaneously heat-up. Another way of saying it is that you cannot build a machine whose sole effects are the cooling of a heat sink and the lifting of a weight.
Huh? Well, in the first case, this ought to match with your day-to-day experience. Let’s say you’ve made coffee upon waking up and then had to help you kids get ready for school, forgetting your coffee on the counter. While the coffee can — and will — cool down if left to its own devices, it cannot — and will not — heat back up to its previous temperature barring some outside intervention.
Why is that? Energy, much like water “seeks its own level,” and it tends to flow from points of high energy into points of lower energy. Picture, if you would, a clear hose which you fill partway with water while holding part of it out of the water. Now put your thumb over the submerged end and hold the two ends of the hose level with one another. Then let your thumb off of the previously submerged side – like so:
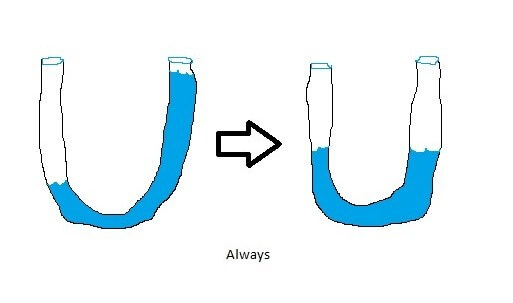
The water always seeks its lowest energy state: it does not spontaneously pile up in one side of the hose. This is the same phenomenon that is on display with your coffee cup.
The other way of describing the Second Law that I mentioned discusses heat and its relation to work. “You cannot construct a machine whose sole effects are the cooling of a heat sink and the lifting of a weight” means simply that you cannot completely convert heat energy into work. That would imply a completely adiabatic (and isentropic) process.
The implication of this expression of the Second Law is that energy has a Quantity (covered by the First Law) and a Quality (the realm of the Second law.) The Quality of energy has to do with how much work which you can extract from it. Entropy (the general implication of the Second Law) is a measure of the energy in a system which can’t be converted into useful work.
So how can this information be put to use in your life? Much like internet ads that promise you limitless energy for your home for free, occasionally you’ll hear stories about somebody who has finally come up with a perpetual motion machine – a machine that will continuously function without energy input in the same fashion forever. Think of a fan blade inside of an evacuated glass bell jar which spins and spins eternally:
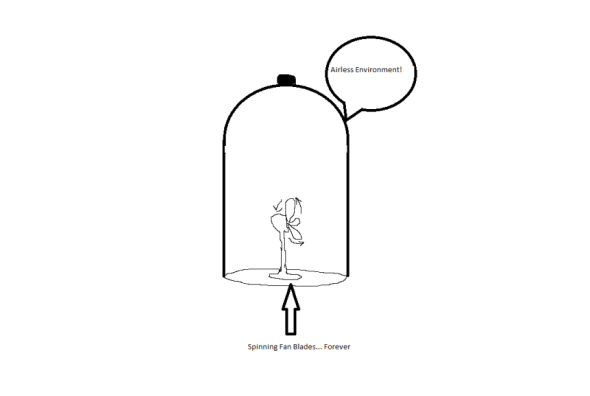
Obviously, this is fiction, as you would need a frictionless connection between the fan blade and the machine. Nonetheless, such things are still sought after.
You might be wondering, how does this inform my worldview? Well, very simply, using these concepts — something doesn’t come from nothing; order in a closed system eventually becomes disorder; etc. — you can begin to assess the truthfulness of some of the wilder claims which you might be confronted with.
From where did the energy powering a device or process come? How is it affecting the environment? Do your suppositions about that violate one of the laws? Keeping those things in mind will banish a significant amount of balderdash.
Published in Religion & Philosophy, Science & Technology



“Of course, there is a lot of confusion about what it is that engineers do.”
Don’t be silly. They drive the trains!
Saturday Night Science began on 2013-03-09 and concludes, after 103 episodes, with this post.
Saturday Night Science: Rockets and People. Vol. 4
anonymous
April 25, 2015 at 4:37 pm ( 3 weeks ago )
14 comments
15 followers
Maj–
.. and that spinning energy can be very quickly translated into an increase in temperature in certain conditions.
Is this for a later post because I’d love to hear about this?
I don’t know if it’s a thing that is large enough for its own post, but it works like this:
What Midge was alluding to when she was talking about “Statistical Mechanics” is a statement of fact. The thing that we conceive of as “temperature” with something like air is really an approximation; an average. In reality, when you get down to the most granular level, at the scale of the atomic the thing you experience as air temperature is a phenomenon which is comprised of a lot of particles zooming around at different speeds with quite different individual energies.
The temperature of a particular molecule is defined by how much kinetic energy it has; this is expressed in a very real sense by the fact that that particle vibrates. The higher the temperature, the more vigorous the vibration and the higher the kinetic energy the particle has.
If you’re following me so far, imagine a water molecule as you would a baseball. Its temperature is a function of its energy; the higher the energy the higher the temperature and the more vibration AND the higher the velocity it has. In addition to this, the baseball could be spinning very quickly. So, when you’re calculating the net kinetic energy of the baseball/molecule you have to account for its velocity and its rotational energy.
CONT’D
The boiling water thing occurs when you microwave water molecules – the microwaves affect the molecules in a couple of different ways. They add to the net kinetic energy of the molecules by increasing their temperature (their straightline velocity) but the waves can also “spin” the molecules such that their net energy is actually above that required for vaporization – they do not have the appropriate straightline velocity to “escape” their liquid state and begin to boil, but the sum of their straightline and rotational energies is higher.
The issue comes when you introduce a nucleation medium into otherwise relatively pure, superheated water which causes that rotational energy to be quickly converted into straightline velocity – this can result in an explosive reaction.
Picture if you would, a tire on a dynamic balancing machine. The tire is spinning very quickly, but doesn’t go anywhere because it can’t find purchase – nonetheless it has immense rotational energy. If that tire were to make contact with the ground it would zoom off in the direction of its rotation. This is like the molecules in superheated water.
http://www.snopes.com/science/microwave.asp
I don’t remember where, now, but I once saw the three laws of thermodynamics succinctly summarized by a physicist for laymen (perhaps a bit tongue in cheek…) as follows:
1. You can’t win.
2. You can’t even break even.
3. You can’t get out of the game.
As a nuclear engineer once said to me:
The Laws of Thermodynamics
And people wonder why engineers are pessimistic…
Are you saving the Third Law for another post?
The Third Law of Thermodynamics is that no object can have an absolute temperature equal to zero.
Now, you may well be asking why winter temperatures that drop below zero are not a violation of this law. Well, that’s because C and F are not absolute temperature scales!
As Majestyk mentions above, temperature is the average speed/energy of motion of molecules in a material. Think of measuring the speed of all the cars in a city (city leaders are probably drooling at that) and finding how much they move on average. Molecules in a solid are like me trying to parallel park – going back and forth over the same area. The natural starting point is zero motion – everything is at rest. That is what we call absolute zero, and it is the starting point for the Kelvin (metric/SI) or Rankine (USA!) system. Absolute zero is −459.67°F (0R) / −273.15°C (0K) , so you can see why we use a different zero. Absolute temperatures are required for thermodynamic math.
Now, you may wonder about negative absolute temperatures. They are very strange: hotter than the same positive temperature, and key to the operation of lasers. But my lunch break is over &I am out of space.
I know that you’re joking, but there is a serious point to be made. Physical laws apply to physical things; they do not apply to ideas. Arguing, say, that the Second Law of Thermodynamics means that nations must eventually “run down” and fail is a fallacy. Nations are not physical things; they are ideas and their success or failure has nothing to do with Thermodynamics. People all too often try to apply scientific theories (e.g., Relativity and Evolution) to areas – such as society – in which they make no sense.
You did this on purpose didn’t you?
What a cliffhanger.
Shoulda just boiled it down in song form:
No it isn’t, as long as you use a long enough timescale.
Since,eventually, the sun will expand to envelop the Earth, thereby destroying all vestiges of life, and since, eventually, entropy will cause the Universe to succumb to heat death, it therefore follows that all nations indeed must eventually run down. Can’t have a nation without life, after all.
When in high school, my AP US History teacher called a parent conference to discuss my troubling class performance. I used the 2nd law of Thermodynamics (entropy: a system goes from order to disorder) to explain why my homework submissions began to decline in quantity and quality as the semester went on.
It didn’t go over that well.
I would state the Third Law as follows: The entropy of a crystal as it approaches absolute zero also approaches zero.
The reason being that no structure is ever truly free from disorder and things like quantum fluctuations introduce imperfections. When you hear scientists discussing the imperfections which are visible in the Cosmic Background Radiation this is what they’re talking about. Even at the universe’s earliest, least entropic state the quantum fluctuations (responsible for the structures that we see in the universe currently) introduced imperfections in the cosmos’ structure which are still evident today.
The most common example of this sloppy thinking is the application of Newton’s Third Law to politics: Every action has an equal and opposite reaction. Try googling it, you’ll see it used as a lazy explanation for all kinds of political turmoil.
But in this case, it’s accurate. The Keynsian Multiplier is a fantasy, there’s no evidence that it exists.
Fair point, but too often that’s because people don’t understand the laws or they apply them poorly.
But we do have a word for ideas which do not comply with the physical laws: fantasy.
God doesn’t need policemen because His laws can’t be broken.
Tuck,
You’re right that the Keynesian multiplier is a fantasy, but the First Law is not what “proves” that it’s false. Mathematics and a little common sense is enough.
Keynes’ multiplier = 1/(1-MPC), Where MPC is the marginal propensity to consume. If we consume everything produced and imported, then MCP goes to 1 and the multiplier goes to infinity. In other words, all we have to do is eat our seed corn and we’ll be infinitely rich.
It doesn’t take Physics to detect the flaw in this reasoning. It does, however, take a PhD to accept it.
The Wag you are looking for is Ginsberg, and this is his parody on thermodynamics. I use it all the time for folks I am conversing with who never had physics. It is much easier to get the idea across than even what Majestyk relatively short exposition does.
LOL. I was thinking along these lines:
That’s exactly what the multiplier purports to do, as you point out.
Can you point me to more on this? I don’t really understand it.
Also, is this their official formula or just something to approximate their reasoning?
It’s official…
Don’t cry too much when you realize this is how the world is run…
Ah, but there is such a thing as a lunch someone else pays for. Which is close enough for me.
Forgive me for ending a sentence with a preposition. “A lunch for which someone else pays” seemed too pretentious.
I have a couple of comments on hidden assumptions:
I also have a comment about the First Law of Thermodynamics generally. As I understand the current state of cosmology, the entire universe appeared out of nothing, so we are living in one big violation of the First Law. (Of Thermodynamics, I mean, not robotics.)
Contra the diktat of a 17th century Latin grammar fetishist, a preposition is a perfectly fine thing to end a sentence with.
Smart alecks.
It’s Keynes’ official formula. There are a lot of links on this. Here’s one:
http://welkerswikinomics.wikifoundry.com/page/The+Multiplier+Effect
Tuck,
Sorry, just saw that you had already answered Captain Power.
The more the merrier. I am a big fan of curated content.
Even though I could google something myself, to have someone who actually knows what they are talking about look through a list of links and pick that they think is suitable is pretty great. Thanks!
I see. Thanks.