Ricochet is the best place on the internet to discuss the issues of the day, either through commenting on posts or writing your own for our active and dynamic community in a fully moderated environment. In addition, the Ricochet Audio Network offers over 50 original podcasts with new episodes released every day.
 Share Your Expertise: Vintage Perfume and Biochemistry
Share Your Expertise: Vintage Perfume and Biochemistry
One of the ironies of fragrance is that organic compounds used by plants as natural pesticides and toxins (to repel predatory insects and herbivores) are some of the key ingredients in perfumes … which are used by human beings to attract, not repel, other human beings (in theory, anyway). 1
These organic compounds (known as secondary metabolites) are present in many of the essential oils used in perfumes, but their key components weren’t manufactured synthetically until the late 19th century. Along with synthetic molecules created in the lab, advances in chemistry at this time meant that traditional extraction processes could be standardized and mass-produced, resulting in a high quality (and quantity) of essential oils and natural isolates. Oils extracted by traditional small-scale methods varied greatly in quality, and could be sludgy and burnt-smelling due to high contaminant levels.
The availability of synthetic compounds and the use of standardized production techniques meant that perfumers could choose from a much larger selection of raw materials, at a much higher quality and lower cost, than ever before.
Coumarin was one of the first aromachemical superstars. Paul Parquet, perfumer for the French house Houbigant, used it to create the fragrance Fougère Royale (Royal Fern) in 1882. Coumarin is present in Tonka beans (Dipteryx odorata), which have a fresh, grassy scent with notes of hay, almond, and vanilla. The synthetic form of coumarin was isolated from coal tar in 1868 by English chemist William Henry Perkin, and was used in the 1880s and 1890s for perfumes and for flavoring cigarette tobacco. 2
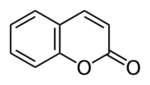
Coumarin molecule
The anticoagulant drug warfarin (trade name Coumadin) is synthesized from dicoumarol, which is formed in nature when sweet clover hay goes moldy in a wet environment and the coumarin in the clover interacts with certain species of fungi. A series of wet summers in the US and Canada during the 1920s, and the resulting moldy clover hay, led to an epidemic of cattle and sheep bleeding to death. After the compounds responsible for the hemorrhaging were isolated, warfarin was patented as a rat poison … and as a blood thinner in humans.
In humans, dermal use of synthetic coumarin (as in perfumes and cosmetics) is safe. And essential oils containing natural coumarin have never caused any adverse reactions when used on the skin. 3 So no need to panic when you see coumarin or Tonka listed as a perfume ingredient!
Houbigant’s Fougère Royale was a ground-breaking scent that became wildly popular. It created the fougère family of masculine fragrances, which is still going strong today. 4 Traditional fougères contain notes of citrus, lavender, coumarin, geranium, and oakmoss. They’re fresh and bracing when first applied, and then become richer and deeper with mossy-earthy facets from the oakmoss and hay-almond notes from the coumarin.

Houbigant Fougere Royale
Sharp, spicy aromatic fougères were very popular for men in the 1970s and 1980s – think Fabergé Brut, Azzarro pour Homme, Yves Saint Laurent Kouros, and Guy Laroche Drakkar Noir. Davidoff Cool Water added aquatic and ozone notes to the classic fougère formula. There are scads of Cool Water knock-offs; any men’s fragrance that’s colored blue and has “Sport” in the name is almost certainly a Cool Water wanna-be (my advice is to avoid these like the plague).
I was fortunate enough to run across a bottle of Houbigant Fougère Royale eau de cologne that dates back to the 1950s. It was still sealed when I bought it, though about three-fourths of the fragrance had evaporated. The citrus notes had disappeared almost entirely (citrus oils are very volatile and don’t last long), but the lavender was still cool and minty, and the coumarin-oakmoss base was deep and complex with hay, vanilla, toasted almond, and tobacco facets. The overall effect is rich but never sweet or cloying. It’s a lovely masculine scent, very classic and poised. Unfortunately the modern version of Fougère Royale lacks the deep, rich base notes of the vintage formulation, due to recent limitations on the use of oakmoss in fragrances.
Other aromachemicals that took the perfume world by storm at the turn of the 20th century include vanillin (synthetic vanilla; Guerlain Jicky), eugenol (spicy clove-carnation; Roger & Gallet Blue Carnation), and C-14/gamma undecalactone (ripe peaches; Guerlain Mitsouko).
The most famous perfume of all, Chanel No. 5, gets its shimmering, glittering texture from three aliphatic (fatty) aldehydes – C-10/decanal (waxy orange rind), C-11/undecanal (clean and “perfumey”), and C-12/lauric aldehyde (clean waxy floral). These aldehydes were used at very high levels by Ernest Beaux, who composed No. 5, as a way to lighten and enhance the gorgeous rose-jasmine accord at the heart of this perfume.
Vintage Chanel No. 5 also contains nitromusks – which are byproducts of TNT (trinitrotoluene, the explosive) that were discovered by German chemist Albert Bauer in 1888. These musks have a rich, intense, animalic smell that’s characteristic of many mid-20th-century perfumes. When some nitromusks were found to be phototoxic (causing allergic reactions on skin when exposed to sunlight), their use was discontinued. 5 Modern Chanel No. 5 is nitromusk-free, and doesn’t have the rich animalic base notes of the vintage formula … so if you have a bottle of old-school No. 5 that belonged to your mother or grandmother, treasure it!
What vintage perfumes have you worn, or do you remember your parents or grandparents wearing?
________________________
1 There are always exceptions … it could be argued that Axe is in fact a type of scent-based pesticide designed to repel anyone with a functioning olfactory nerve.
2 Simon Garfield, Mauve: How One Man Invented a Color that Changed the World (2000), 173.
3 Robert Tisserand and Rodney Young, Essential Oil Safety, 2nd ed. (2014), 544.
4 Derek B. Lowe, The Chemistry Book: From Gunpowder to Graphene, 250 Milestones in the History of Chemistry (2016), 176-7.
5 Charles S. Sell, ed., The Chemistry of Fragrances, 2nd ed. (2006), 96-8.
N.B.: Both images were found on Wikimedia Commons, and both are in the public domain. The Fougère Royale image is courtesy of the Osmotheque.
Published in Group Writing




Love your lede!
I had thought about doing a post on gardening, but this summer was a disaster garden-wise! The weather here in Michigan went from drought to monsoon rains, and the Japanese beetles devoured almost everything I planted that survived the floods a few weeks ago.
So vintage perfume it was …
Thanks, Titus!
The whole post is great, but this really made me laugh.
Well, you know what they say about the weather here, if you don’t like it, wait a few minutes.
A genuinely fascinating look at something I know nothing about…one of Ricochet’s greatest pleasures!
I once bought cologne from Kiehl’s, one of the oldest pharmacies in Manhattan, a strange and ancient apothecary on the lower east side. They did some basic formulation right there in the shop, and kept everything in glass bottles custom made to fit a floor-to-ceiling cabinet of varnished wooden drawers, each with its name handwritten in elaborate script.
Thanks, Anna M!
Despite being a child of the Sixties, my wife has always worn a legendary ’30s perfume, Shocking by Shiaparelli. I even bought her the famous original bottle, designed by Salvador Dali and shaped like a dressmaker’s dummy with a bouquet of flowers for a head. She has enough to last the rest of her life — a good thing because the latest incarnation of Shocking seems to have changed the recipe in a noticeable way.
As for me, the fragrance is literally intoxicating. My face flushes, I have a hard time breathing for a few seconds, and it evokes a lifetime of memories (and, despite the Dali connection, not all of them surreal).
The one perfume I remember a lot of women wore in my childhood (besides Chanel No. 5 and Joy) was Tabu. Nasty, cloying stuff. My mother never wore it because my dad hated it. My father, a spy and a rather worldly man, always said that Tijuana whores almost bathed in the stuff. I never asked him how he knew that.
Regarding Axe: If you really want to know how horrible that stuff is, spend a day at a Boy Scout camp. My Scouts use endless quantities of that evil concoction as a deodorant instead of taking showers. They also spray Axe deodorant on bugs and set them on fire when no adult is looking.
Cinnabar (from my grandma) and a very old bottle of Tabu, a first-formulation Samsara. Also older versions of men’s fragrances.
Are you a chemical engineer by training, Anna? A professional perfumer? A hobbyist? Do you know about Bo Jensen’s site? Bois de Jasmin?
I am – or was – a would-be perfume hobbyist, though respiratory problems mean avoiding it for long stretches (maybe forever, if I knew what was good for me). I once maintained a small library of raw perfume ingredients, mostly essential oils, in an attempt to smell nice things while avoiding mixes containing unknown irritants.
@vicrylcontessa is a more successful perfume hobbyist than I.
You’re welcome, Gary! Kiehl’s Musk is famous among perfume fanatics as one of the best “skin perfumes” from the 1970s. The vintage formula has a soft, powdery clean-skin scent that layers beautifully with florals. Kiehl’s still sells it, but it’s a shadow of its former self (like so many classic fragrances).
Kiehl’s still uses the traditional amber Boston round bottles; that design dates back to 19th-ct apothecaries. The floor-to-ceiling cabinets in the Manhattan store sound impressive!
One I remember a lot of women wearing is L’air du Temps. I think it’s still made. One I HATE is White Shoulders.
I use Design by Paul Sebastian, and I regularly get people telling me I smell good, or asking me what I’m wearing, and once a sales clerk *told* me what I was wearing. She was right.
One question. Why are the perfumes made by Estée Lauder able to detected from so far off? I swear you can smell the people wearing them a good 50 feet away, and that’s not a good thing.
Very interesting post.
Vintage Schiaparelli Shocking is gorgeous – roses and honey with a touch of spice. It can turn if not stored carefully (someplace cool and away from light; never in a bathroom), but when it’s well-preserved it really is intoxicating. What a great present for your wife!
And yes, Dana Tabu is a tad overwhelming, isn’t it! Patchouli and animalics aren’t exactly subtle …
Horticulture’s loss is our gain. Great post, thanks.
I do know what you mean about the garden. We went from monsoons to drought, to monsoons again. And it was the 17-year locust cycle in Western PA, which was the worst I’ve ever seen or heard–deafening. They did keep the Japanese beetles down, or at least it seemed that way. As soon as the locusts died off, though, the beetles moved in.
“Nice perfume, lady, but must you bathe in it?”
Huh. Perhaps one reason I don’t find Tabu off-putting is I never ran across those who did bathe in it. I have run across colognes intended for bathing – folks used to bathe their temples in 47-11 to relieve headaches and summer heat, apparently. I have my grandma’s bottle of that, too.
Odd bit of chemistry. Apparently estratetraenol (secreted by women) smells like cedarwood and androstenol (secreted by men) smells like sandalwood – specifically, it’s the “sandalwood odorant with the lowest odor threshold and even serves as template for the sandalwood olfactophore”. The cedarwood smell is often considered more “male” than the sandalwood smell, though. (Also, Australian sandalwood reminds me of freshly-cooked pasta for some reason, mildly curious to know why.)
I fell down the rabbit hole (as perfumistas say) about four years ago, when I was looking for a new perfume to replace my old standbys Clinique Happy and Balmain Vent Vert. Now I’m reading biochemistry textbooks and thinking of starting my own perfume blog! It’s a
consuming obsessionserious avocation for me now.I’m sorry to hear about your respiratory allergies! I dread even catching a cold now; I hate not being able to smell anything.
Bo Jensen and Bois de Jasmin (by Victoria Frolova, who works in the fragrance industry) have two of my favorite perfume blogs. Now Smell This and the Perfume Posse are excellent as well.
I was thinking of Vicryl Contessa when I was putting together the post!
Thanks, I’m glad you liked it!
Most major perfume houses use pre-existing bases, or signature accords, in their fragrances so they don’t have to re-invent the wheel every time they create a new perfume. This practice also creates a continuity and a distinctive feel for a house, which customers recognize and learn to like (or dislike!).
The best example of a signature accord is Guerlinade, a combination of rose, jasmine, Tonka, and vanilla that’s been used by the house of Guerlain since the late 19th ct. They even released it as a perfume on its own in 1998.
Lauder has a definite signature accord, and quite a few perfumistas don’t like it either (I’m one of them). It smells synthetic and rancid to me, and it’s horrible with my skin chemistry. But Lauder does have a dedicated following who think “Lauderade” is wonderful. Market forces in action, I suppose!
Sublingual immunotherapy is becoming more common for respiratory allergy desensitization. As it becomes more widely-known and better-understood, it wouldn’t surprise me if more folks started DIYing it and publishing how-to’s with their results. (I would love to be not-allergic to oakmoss, for example.) Desensitizing oneself to easily-avoidable cosmetic ingredients is understandably viewed as a frivolity, but would be easier to do on one’s own, one would think, than collecting pollen, mold spores, dander, etc for titration.
So, you put the perfume under your tongue?
I tried that with my cats, but they really, really didn’t like it.
Love my grandma’s old bottle of Cinnabar. Can’t remember liking any other Lauder product I smelled. Disposed of one bottle I received by pouring it over mounds and mounds of Epsom salts, then giving it away to female relatives as a “spa treatment”. They seemed to enthusiastically like it, which is great, because I wouldn’t.
A sufficiently diluted mix of whatever you’re allergic to. (How DIYers should determine “sufficiently” is TBA.)
Did you dilute your cats enough before attempting?
Unfortunately people who like to over-apply their fragrances are usually the ones with the worst taste [sigh].
It’s like people who play their car stereos at ear-bleeding levels … the loudest music is always stuff you don’t want to hear.
But, but … Egon Spengler would approve!
They don’t really like my attempts at diluting them.
And I suppose bathing them in alcohol or hexane to better extract their volatile compounds wouldn’t help!
How have we not yet promoted this to the main feed! Help, folks!
Great post. Enjoy it!
My grandmother used Millot Crêpe de Chine she received as a gift in the mid-1950s.
Good point. Now at 6.
If we’re nice, the lady might stay & tell us more! Get the gang to comb their hair & take off their caps-